Stryker
In the summer of 2023, I completed an internship with Stryker Sports Medicine in San Jose, CA as an R&D intern. I had the opportunity to work on the designing, prototyping, and testing processes of multiple surgical devices in all stages of development. I was lucky to be able to collaborate with many different teams and engineers and complete meaningful work toward the development and release of products for Stryker. I worked at a high level both hands-on and digitally, using machine shop tools and polymer 3D printing, operating in mock surgical conditions, utilizing Solidworks and Solidworks PDM to create complex CAD files and assemblies, and working with both legacy and new designs.

Projects Summary
Over three months, I worked primarily on four projects, two of which were centered on new product design. The first was a snap-on fixture that could be optionally added to a production surgical device, with the need for this product coming from surgeon feedback and competitor products in the market. The second new product design project was a ground-up project of a new surgical tool. I worked as the lead designer for this product, working with the marketing department as I designed and prototyped. The other two projects I worked on were product development testing. One of these products was in the late stages of development, getting ready for release, and the other was a product in the early stages, starting testing.
Product Design

Snap-on Fixture
As a medical devices company, Stryker is always innovating and iterating their surgical tools based on new research, new surgeon needs, and new technologies, all focused on creating easier, more successful surgeries. While this often means creating new products to replace old offerings, occasionally changes can be made to products through external additions. I was tasked with designing, prototyping, and testing a snap-on fixture for a line of products that would increase their usage capabilities. Using primarily Solidworks and GD&T, I designed a variety of snap-on fixtures that would have the physical properties necessary to achieve the desired results. Through iteration and prototyping using an inkjet 3D printer, I made and tested multiple different options with a wide range of shapes. Through this iteration process, I was able to pinpoint the most effective design for the desired results, and through testing confirmed adequate, repeatable data. As a result, the snap-on fixture that I designed began usage for the testing process that the product is undergoing. This fixture will allow the product to have expanded use cases and increased safety.

New Surgical Device
As previously stated, Stryker is constantly developing new products to improve performance and fill gaps in existing product lines. Stryker was looking to expand one of these product lines based on a specific type of surgery, in which the current products were working, but could be better. I took on the design project as the lead and began by researching the existing products and the desired surgery to understand where the shortcomings were and what could be improved. I also received marketing’s desires for the product’s design and features. Using this information, I used Solidworks to create a multi-piece assembly of the new product. This assembly had to have a cover piece that could slide in and out of place easily but never fall off. I also had to design within the existing silhouette of a product so that the new product would feel familiar to surgeons and would fit in existing packaging. The largest constraint was the force requirement, as the device had to be capable of withstanding multiple sharp blows. After finishing the CAD, I used an inkjet 3D printer to prototype the product, then completed other assembly through the dismantling of similar products and pieced together a working prototype. With this prototype, I performed testing in a mock surgical environment and from my results, I returned to my CAD to iterate and improve. After a few cycles of prototyping, testing, and iterating, the product was delivered to marketing. As a surgical device, there will be many levels of testing and design modifications before this product reaches the market. However, the first steps are complete and due to the need for this product, it will likely hit the market in the upcoming years.
Product Development Testing

Near Market
An updated product of one of Stryker’s more important lines is nearing release, and some final testing still needed to be done. Over the course of my internship, I spent hours assembling these devices for testing and also ran tests. While assembly can often be seen as simply manual labor, it was a good learning experience about designing for assembly. These devices had to be assembled in a factory before being shipped out to surgeons, so the better they were designed, the faster and easier they could be assembled, minimizing errors. Through assembling these products, I learned about tools for assembly that Stryker uses and also design additions that can help assembly. The tests I ran were primarily UTS tests using hand-held force gauges and an Instron. In a multi-piece device such as this one, it was interesting to learn about options for testing and how to test specific pieces to failure. The data I collected from my testing was constantly recorded and compiled with all test data from the product and affected design changes and product specifications.
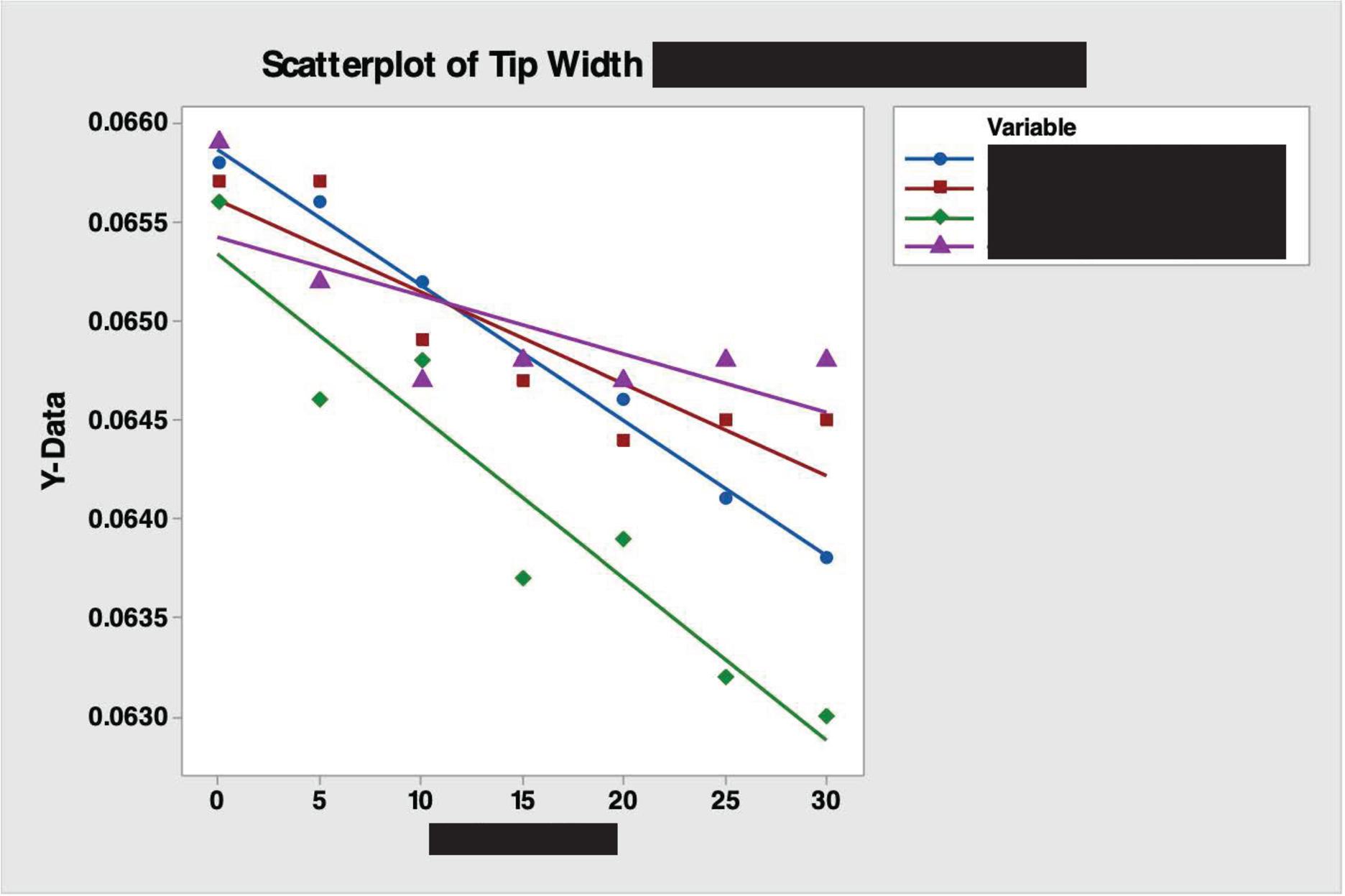
Early Development
I led the first rounds of material testing for a new product in early development. After receiving samples of the product in two different materials, I used the spec. sheet to check whether the product was within the dimensions ordered. I then tested the two materials in a mock surgical environment, specifically watching material wear over a number of cycles. At constant intervals, I used a Keyence imaging system to view the product at high magnification and measure the changes in dimensions due to wear. Overall, I completed this testing for three separate materials with products from two separate production lines and under two different sets of conditions, coming out to a total of five rounds of testing. I recorded all of my data both manually and digitally and wrote a testing report at the end of every set of tests, sharing the report with all engineers involved in the product. Based on my testing, a material was chosen for its superior performance, and the two sets of conditions were proven to both be safe for the usage of the product.
Reflection
My time at Stryker helped greatly to develop my design and engineering skills. I’m very grateful to my team for their help throughout my internship and the friendships I made. My favorite project was designing a new surgical device from the ground up and I enjoyed every failure and learning experience along the way.